Machine Learning Applications in Head and Neck Radiation Oncology: Lessons from Open-Source Radiomics Challenges
July 1st, 2018
Categories: Visual Analytics, Visual Informatics, Machine Learning
Authors
About
Authors: Hesham Elhalawani, Timothy A Lin, Stefania Volpe, Abdallah S.R. Mohamed, Aubrey L. White, James Zafereo, Andrew Wong, Joel E. Berends, Shady Abohashem, Bowman Williams, Jeremy M. Aymard, Aasheesh Kanwar, Subha Perni, Crosby D. Rock, Luke Cooksey, Shauna Campbell, Pei Yang, Khanh Nguyen, Rachel Ger, Carlos Eduardo Cardenas, Xenia Fave, Carlo Sansone, Gabriele Piantadosi, Stefano Marrone, Rongjie Liu, Chao Huang, Kaixian Yu, Tenfei Li, Yang Yu, Youyi Zhang, Hongtu Zhu, Jeffrey S. Morris, Veerabhadran Baladandayuthapani, John W. Shumway, Alokananda Ghosh, Andrei Pöhlmann, Hady Ahmady Phoulady, Vibhas Goyal, Guadalupe Canahuate, G. Elisabeta Marai, David Vock, Stephen Y. Lai, Dennis S. Mackin, Laurence E. Court, John Freymann, Keyvan Farahani, Jayashree Kalpathy-Cramer, Clifton D Fuller
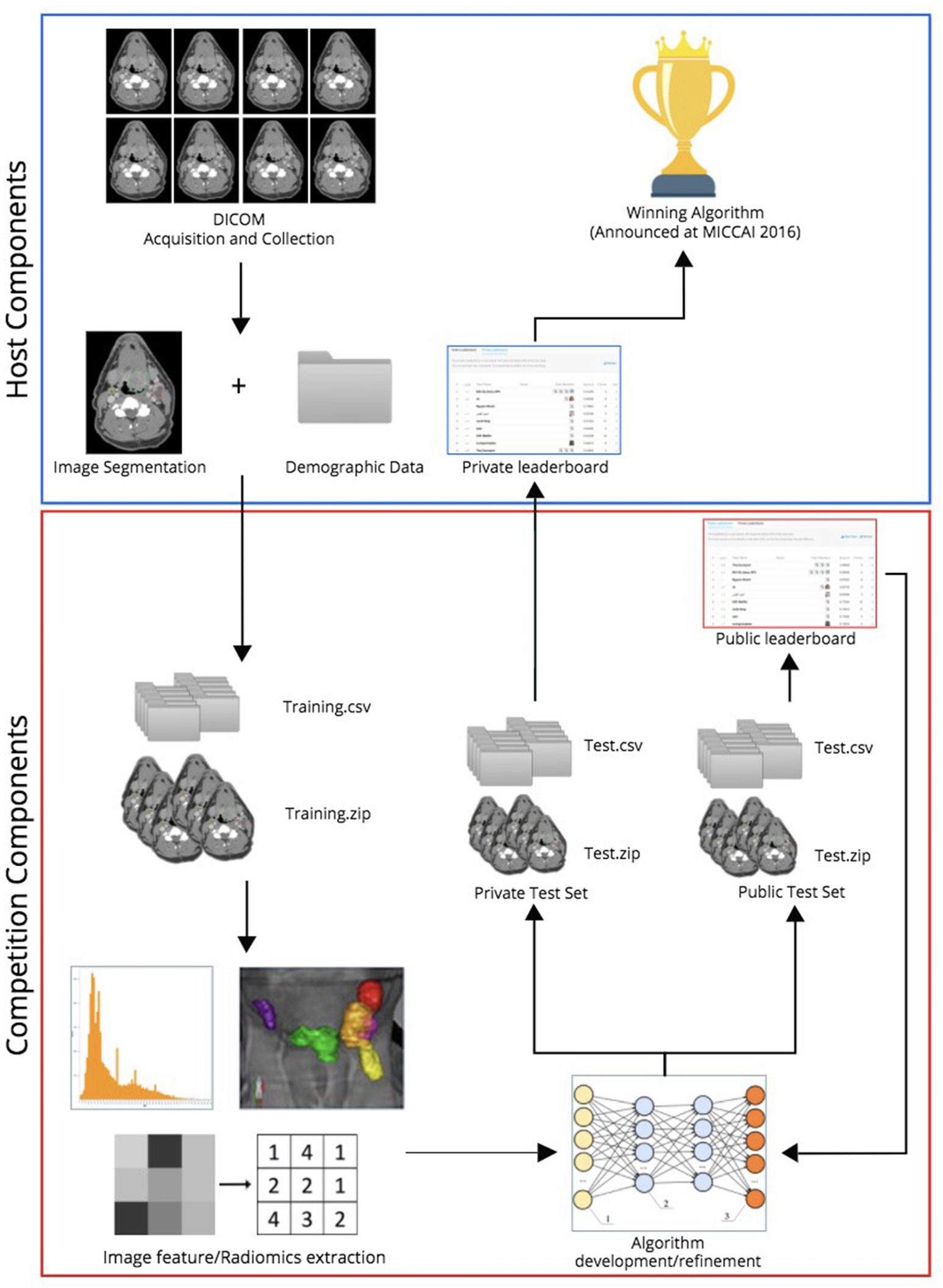
Abstract: Radiomics leverages existing image datasets to provide non-visible data extraction via image post-processing, with the aim of identifying prognostic, and predictive imaging features at a sub-region of interest level. However, the application of radiomics is hampered by several challenges such as lack of image acquisition/analysis method standardization, impeding generalizability. As of yet, radiomics remains intriguing, but not clinically validated. We aimed to test the feasibility of a non-custom-constructed platform for disseminating existing large, standardized databases across institutions for promoting radiomics studies. Hence, University of Texas MD Anderson Cancer Center organized two public radiomics challenges in head and neck radiation oncology domain. This was done in conjunction with MICCAI 2016 satellite symposium using Kaggle-in-Class, a machine-learning and predictive analytics platform. We drew on clinical data matched to radiomics data derived from diagnostic contrast-enhanced computed tomography (CECT) images in a dataset of 315 patients with oropharyngeal cancer. Contestants
were tasked to develop models for (i) classifying patients according to their human papillomavirus status, or (ii) predicting local tumor recurrence, following radiotherapy. Data were split into training, and test sets. Seventeen teams from various professional domains participated in one or both of the challenges. This review paper was based on the contestants’ feedback; provided by 8 contestants only (47%). Six contestants (75%) incorporated extracted radiomics features into their predictive model building, either alone (n = 5; 62.5%), as was the case with the winner of the “HPV” challenge, or in conjunction with matched clinical attributes (n = 2; 25%). Only 23% of contestants, notably, including the winner of the “local recurrence” challenge, built their model relying solely on clinical data. In addition to the value of the integration of machine learning into clinical decision-making, our experience sheds light on challenges in sharing and directing existing datasets toward clinical applications of radiomics, including hyperdimensionality of the clinical/imaging data attributes. Our experience may help guide researchers to create a framework for sharing and reuse of already published data that we believe will ultimately accelerate the pace of clinical applications of radiomics; both in challenge or clinical settings.
Keywords: machine learning, radiomics challenge, radiation oncology, head and neck, big data
Resources
URL
Citation
, Machine Learning Applications in Head and Neck Radiation Oncology: Lessons from Open-Source Radiomics Challenges, Frontiers in Oncology 2018, vol 8, pp. 1-14, July 1st, 2018. www.frontiersin.org